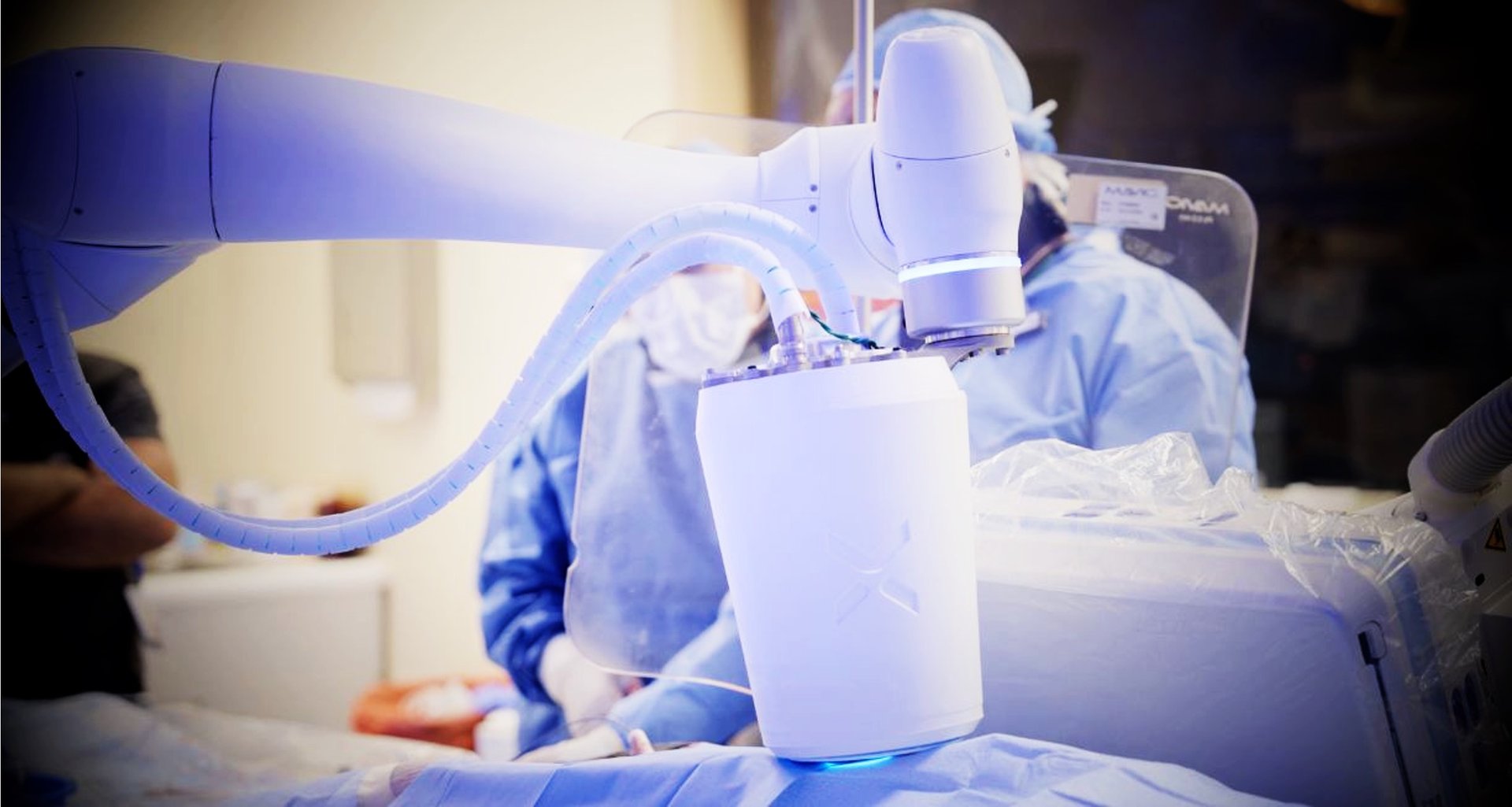
COMMERCIAL ENTRY: LOWER-LIMB CTO
Flux Robotics is focused on complex lower-limb Chronic Total Occlusion (CTO) in CLTI patients as its initial commercial beachhead. We collaborate with leading hospitals and regulatory partners to validate staged clinical expansion.




Clinical & Strategic Partners
COMPLEX LOWER-LIMB CTO (CLTI)
Chronic limb-threatening ischemia affects millions of patients globally. CTO crossing remains the primary technical bottleneck in these procedures.
Flux enables controlled true-lumen navigation, addressing the core unmet need in CTO interventions.


BEACHHEAD INDICATION
REGULATORY STRATEGY
We follow a staged validation approach:
Pre-clinical in-vivo feasibility
GLP validation
First-in-Human CTO study.
This de-risks regulatory entry while building clinical evidence in a high-value indication.


STAGED VALIDATION
PLATFORM EXPANSION STRATEGY
Following CTO commercialization, the Flux magnetic navigation platform expands into fenestration and complex cannulation workflows.
One platform. Multiple high-value vascular indications.



THE FLUX FUTURE
LET'S BUILD THE FUTURE TOGETHER
Get in contact with us to book a demo and explore opportunities.




Controlled Navigation. Predictable Outcomes.
Offices
Groningen
Blauwborgje 31
9747 AC Groningen
Netherlands
Enschede
Ariënsplein 1
7511 JX
Enschede
Netherlands
KvK: 87842165


